Comments
Follow Comments Sorted by time
geluregis
· 7 years ago
· FIRST
The real reason why Edward Elric squeezes his hands together
21
shadowlord
· 7 years ago
You. I like you
7
deleted
· 7 years ago
Damn, that's some serious shit there
4
guestwho
· 7 years ago
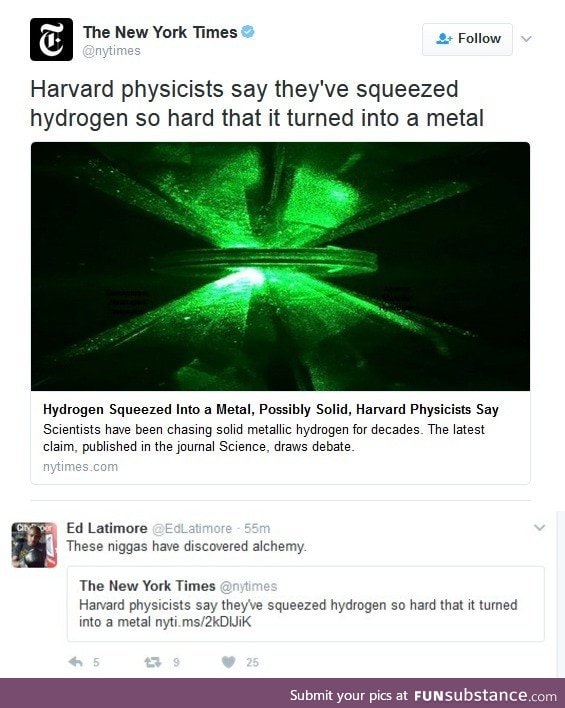
"possibly solid"? Isn't metal definitely solid?
6
gdaymate
· 7 years ago
Mercury is a liquid
4
geluregis
· 7 years ago
A molten metal is still a metal
12
Show All
guestwho
· 7 years ago
OK OK nobody likes a smartass.
▼
skraz
· 7 years ago
nobody likes a dumbass either ;)
15
eternal1
· 7 years ago
Guestwho you uneducated dumbass
guestwho
· 7 years ago
I'll have you know I'm a highly educated dumbass, thank you very much!
eternal1
· 7 years ago
probably educated in other areas
2
guest
· 7 years ago
I think I read the article and said it had metal properties but not solid yet
4
freault4
· 7 years ago
Yes, metallic properties have to do with how the +ions are with the electrons in delocalized bonds (as opposed to ionic or covalent bonds), which can make sense for hydrogen in an *in*organic system. I imagine that this is also why mercury can be a liquid and a metal, because the delocalized bonds can sort of flow over each other. This is also why metals are conducive. *retreats back to corner*
6
matthewg
· 7 years ago
Gaseous Snake confirmed for MGS6!
▼